Welcome to the research projects of Dr. Dirk Becker
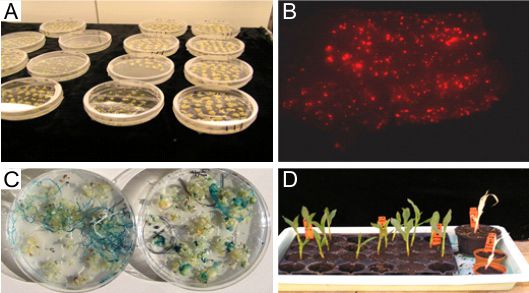
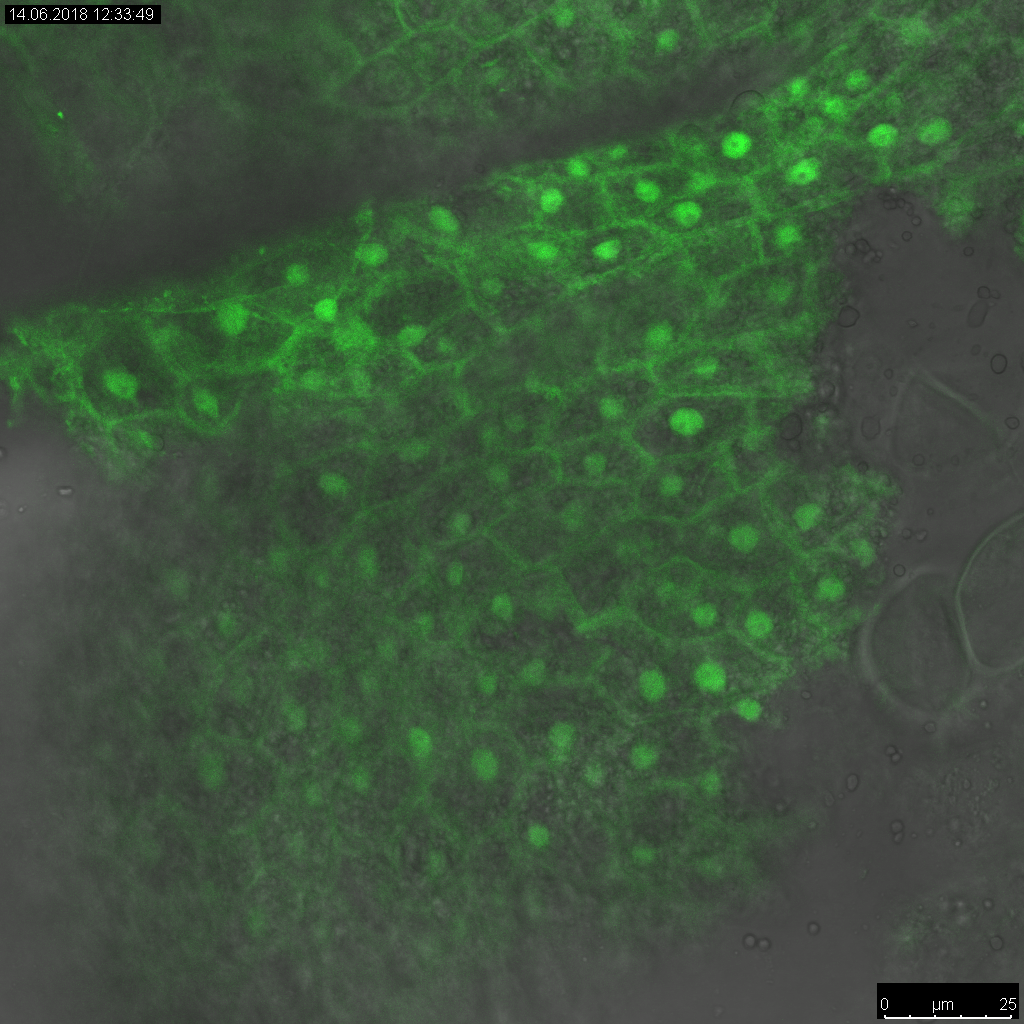
My research group deals with biotechnological and genetic research projects for crop plants with emphasis on cereals, rapeseed and tobacco. Initially, it is necessary to develop in vitro culture systems for an efficient regeneration of plants and, based on that, to establish transformation methods. At present, maize and rapeseeds are routinely transformed in current research projects.
In preceding research projects and by means of these transformation methods we could, for instance, establish starch as renewable resource with defined and partly new characteristics in wheat for the food and non-food sector. Another research focus dealt with the function of storage proteins during dough production of maize and wheat and experiments to improve the baking properties of maize meals through transfer of wheat storage proteins in maize. Additionally, approaches to improve fungal resistance in wheat and millet on transgenic plants were examined.